Once again, the year is drawing to a close, and once again it has been a very exciting and successful year for us. I am pleased to announce that, effective January 1, 2019, we will officially change our name to MEDtech Ingenieur GmbH. We have consistently implemented this step this year, setting the course for the future. This step is the logical consequence of our growth and will support our continued success in the future.
I would like to sincerely thank all our employees, partners, readers, and customers with whom we grow and help make the world a little bit better with good medical products. Of course, we will continue to maintain our blog as usual next year and even include a few surprises.
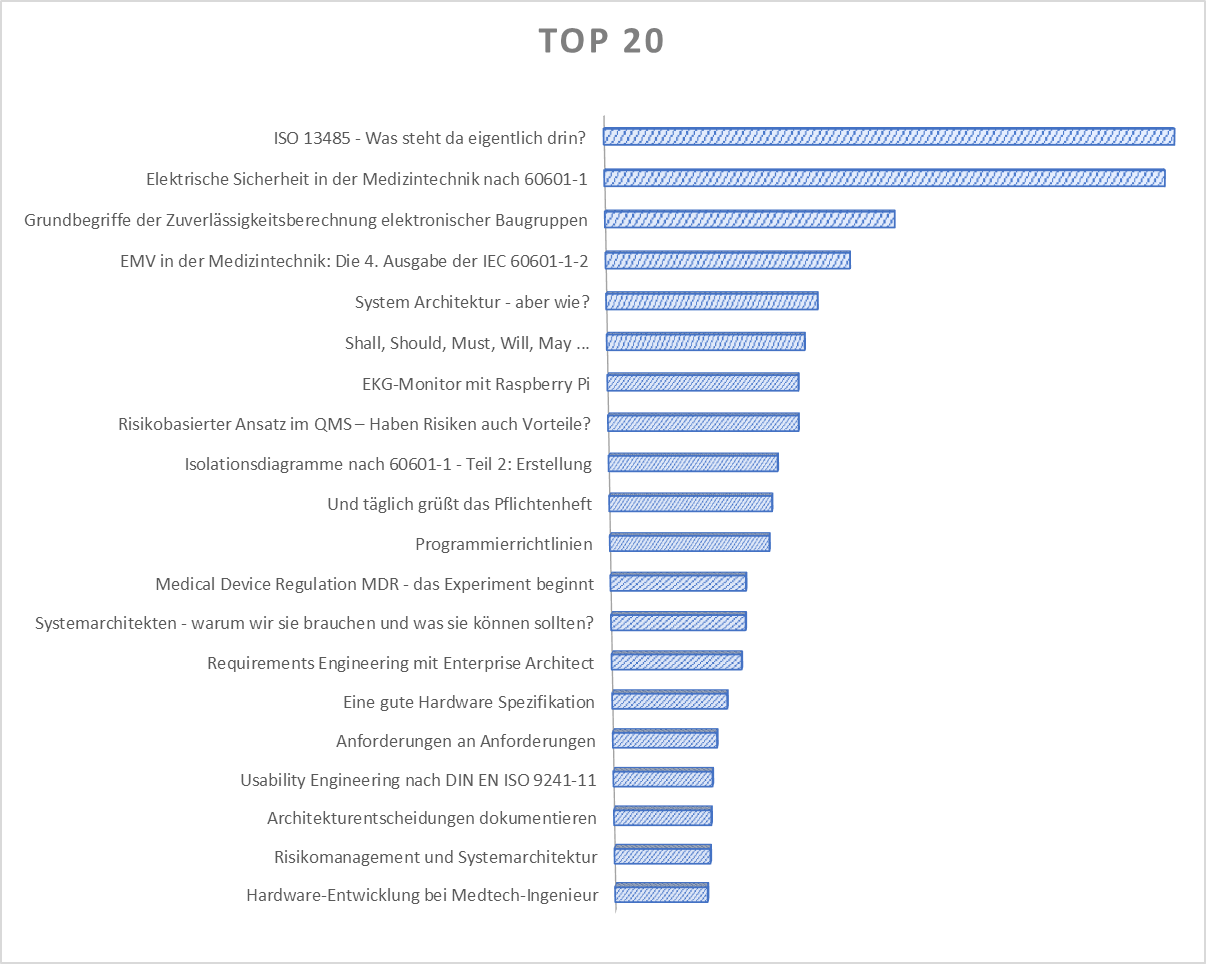
There's been a lot going on on our blog again, and I'd like to share some statistics about our articles. Maybe there's one you don't know yet. Here are our top 20!
We're also active on Twitter and are happy to have many followers. A big thank you to all of our followers!
And for those who don't like reading but prefer watching videos, we've put a few videos online this year. You can find the top 5 here.
Kind regards
Goran Madzar