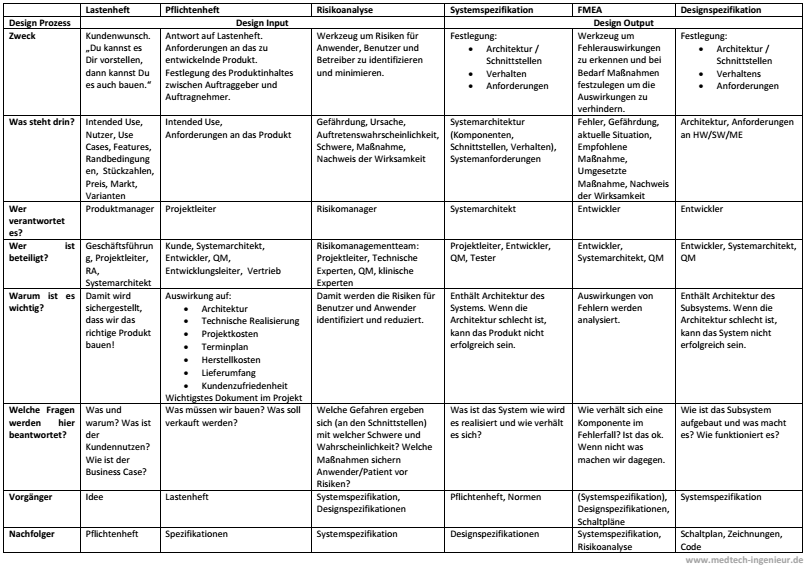
In medical technology, there are a whole series of documents containing requirements. This sometimes leads to a situation where the purpose of each document is unclear, and it's difficult to distinguish between them. To provide some clarity, I've created an overview and highlighted the key design input and design output documents.
|
|
| Dipl.-Ing. Goran Madzar, Partner, Senior Systems Engineer E-mail: madzar@medtech-ingenieur.de Phone: +49 9131 691 240 |
|
Do you need support with the development of your medical device? We're happy to help! MEDtech Ingenieur GmbH offers hardware development, software development, systems engineering, mechanical development, and consulting services from a single source. Contact us. |
|
The result is an overview in table format. It's quite possible that the documents in your organization are named differently, are missing entirely, or that you use additional documents. If so, you can use the table as inspiration and adapt it to your organization. Discuss the table with other people in your company. You'll be amazed at the insights you gain and the confusion that may emerge :-)
To the PDF: Meaning and purpose of the requirements documents
I welcome feedback and would love for you to contact me. Feel free to leave a comment on the article. If you know someone who might also be interested in the blog, I'd be very happy if you would recommend it.
Have lots of fun with it
Goran Madzar