We have already described the basics and the necessity of insulation diagrams for documenting electrical safety according to 60601-1 in several articles:
Electrical safety in medical technology according to 60601-1
Insulation diagrams in medical technology
Isolation diagrams according to 60601-1 Part 1: Basic concepts of insulation coordination
Insulation diagrams according to 60601-1 Part 2: Preparation
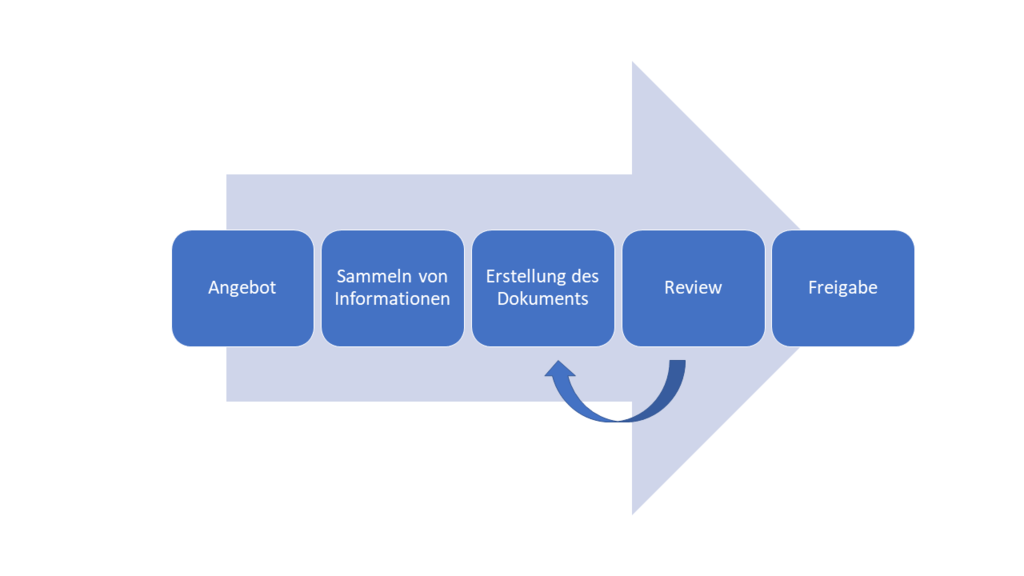
In this article, I'd like to show you our process for creating an isolation diagram for you. The process includes the following steps:
What are your advantages?
- Your employees can focus on other issues that may be more urgent or important.
- Maybe you don't have anyone who wants to delve deeply into the many aspects of 60601-1.
- You will receive an external reviewer for the electrical safety of your system.
- We usually provide a fixed-price quote, which allows you to plan ahead for the effort and costs.
What do the individual steps contain?
1. Offer
If you need assistance with creating insulation diagrams, please contact us. We will discuss the price and timeline with you. We usually offer a fixed price, as the standardized process makes the effort easy to estimate. In addition to creating the entire insulation diagram, we can also provide a review or consultation for you.
| Dipl.-Ing. Martin Bosch, shareholder, hardware developer E-mail: bosch@medtech-ingenieur.de Phone: +49 9131 691 241 |
|
Do you need support with the development of your medical device? We're happy to help! MEDtech Ingenieur GmbH offers hardware development, software development, systems engineering, mechanical development, and consulting services from a single source. Contact us. |
|
2. Collecting information
Once the order is placed, we collect the data necessary for production. Depending on which documents are already available, this data can be found in the requirements specification, specifications, design specifications, block diagrams, etc. The necessary information is described in the article Insulation diagrams according to 60601-1 – Part 2: Preparation and include protection class, input and output signals, classification of applied parts, etc. In addition, there are further requirements and exceptions that can be found in the special specifications 60601-2-XX.
3. Creation of the document
Once we have the information, we can create a diagram, define the routes, and create and fill in the tables according to 60601-1.
Further chapters in the document include an introduction with a description of the system, a description of the requirements and any exceptions.
4. Review
Your staff will then review the document. They know your system best and may notice missing parts, such as metallic, touchable parts, that weren't listed in the original information.
5. Adjustments
Changes may still arise from the review. The document will then be adjusted and expanded accordingly.
The revised document is then reviewed again. If everything is consistent and understandable, it proceeds with approval.
6. Release
Once the diagram is approved, it is signed, thus completing the initial version. During approval, questions, requirements, or comments may arise from the notified body for the medical electrical device, which may require revision of the document.
You can also use the process for your internal creation of isolation diagrams.
If you need support or have any questions about this topic, please call us or send us an email.
We are happy to support you in your medical device development.