Anyone working in the medical technology sector has probably already heard of the ISO 13485 standard. It covers quality management systems for medical devices. But what does this standard actually contain? In this blog post, I'd like to take a look at the standard and provide you with a brief summary of its contents.
ISO 13485 largely corresponds to the old ISO 9001. Since the last update of ISO 9001, the two standards have differed significantly in structure. ISO 9001 is a widely used quality management standard. It is not uncommon for even small tradesmen or their auto repair shops to have an ISO 9001 certificate hanging on their walls. ISO 13485 is specifically aimed at organizations or companies that manufacture or distribute medical devices. The ISO 13485 standard has been harmonized with the EU Medical Device Directives 93/42/EEC, 90/385/EEC, and 98/79/EEC. This allows an organization or company to have its quality management system certified according to ISO 13485 by a notified body and thus place medical devices on the EU market.
The basis for this international standard is a process-oriented approach. This involves introducing, documenting, implementing, and maintaining the processes of an organization or company. The method known as "Plan-Do-Check-Act" (PDCA) can be applied to these processes.
The path to the QM system |
|
|---|---|
Do you need support setting up or maintaining your QM system? Our process will help you achieve your goals quickly:
|
|
Your contact person: |
|
| Dipl.-Ing. Goran Madzar E-mail: madzar@medtech-ingenieur.de Phone: +49 9131 691 240 |
|

The core objective of ISO 13485 is product safety. Product safety is to be increased through compliance with product requirements and the effectiveness of processes within the organization.
In the following figure, I've briefly summarized the contents of the standard. This summary is useful for reminding yourself of the standard from time to time without having to read it in its entirety.
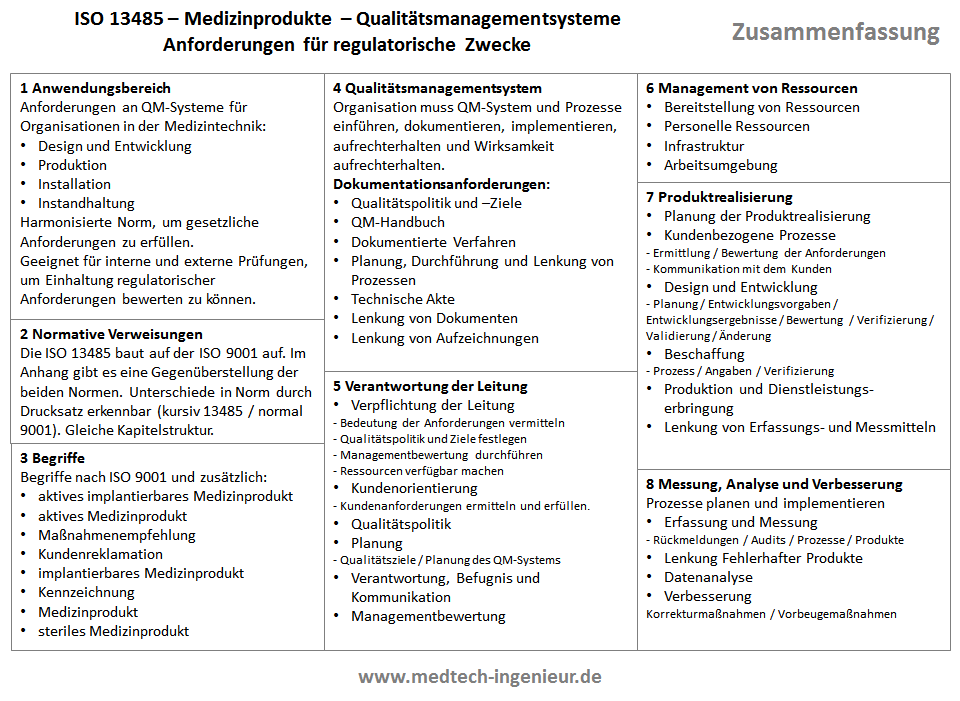 You can see the Summary ISO 13485 also available for download as PDF.
You can see the Summary ISO 13485 also available for download as PDF.
I welcome feedback and would love for you to contact me. Feel free to leave a comment on the article. If you know someone who might also be interested in the blog, I'd be very happy if you would recommend it.
Best regards
Goran Madzar
