IEC 60601-1 is the standard for general requirements for the safety and essential performance of medical electrical equipment. Anyone involved in the development of medical electrical equipment, whether as a quality control engineer, project manager, or developer, should be familiar with the basic structure of the 60601 family of standards.
IEC 60601-1 is an international standard published in Europe by CENELEC as a European standard and then published as EN 60601-1. The European version is published in Germany as DIN EN 60601-1. In the following, reference is always made to the IEC version.
The 60601 family of standards consists of the basic or general standard, the supplementary standards, and the specific specifications. The figure shows the relationship between the various standards.
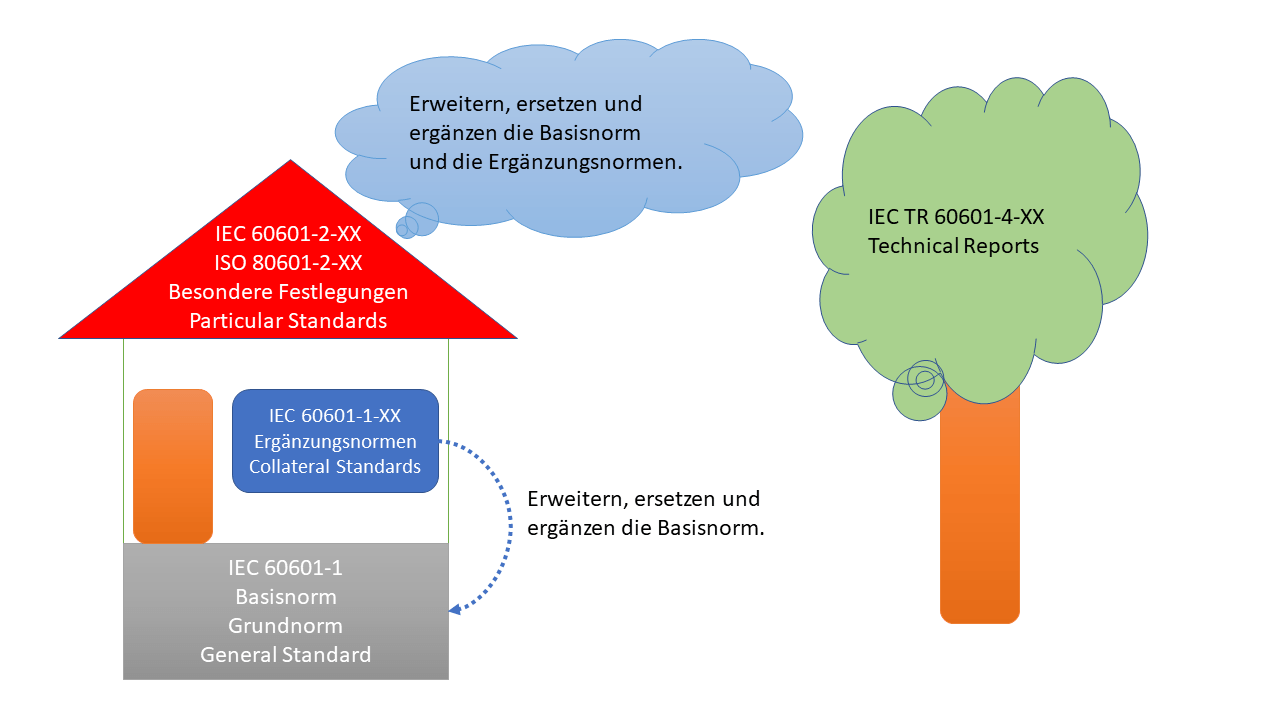
What types of standards are there in the 60601 family?
As shown in the graphic, there are three types of standards. These are supplemented by technical reports.
Basic standard
The basic standard is IEC 60601-1. Other names for the basic standard include general standard, general specifications, basic standard, and general standard. This contains the general requirements for the safety of electrical medical devices.
A revised version of IEC 60601-1 will soon be published as Amendment 2. The IEC currently specifies July 2020 as the expected publication date.
Supplementary standards
The collateral standards are published under the designation IEC 60601-1-XX. These standards contain general requirements for specific groups of medical electrical equipment that go beyond the requirements of the basic standard. For example, the IEC 60601-1-8 standard defines general requirements for equipment containing alarm systems.
| Dipl.-Ing. Martin Bosch, shareholder, hardware developer E-mail: bosch@medtech-ingenieur.de Phone: +49 9131 691 241 |
|
Do you need support with the development of your medical device? We're happy to help! MEDtech Ingenieur GmbH offers hardware development, software development, systems engineering, mechanical development, and consulting services from a single source. Contact us. |
|
The requirements of the supplementary standards supplement, extend or replace the requirements of the basic standard.
Special provisions
The specific specifications are published under the designation IEC 60601-2-XX. These standards contain detailed requirements and tests for specific products or features of medical devices, e.g., IEC 60601-2-4 for defibrillators. The requirements of the specific specifications supplement, extend, or replace the requirements of the basic standard and the collateral standards.
There are also special specifications developed jointly by ISO and IEC committees. These are known as ISO 80601-2-XX published.
Technical reports
For explanation and guidance on implementation, so-called Technical Reports are published for some standards or topics under the designation TR 60601-4-XX, e.g. IEC TR 60601-4-2 with the title "Medical electrical equipment - Part 4-2: Guidance and interpretation - Electromagnetic immunity: performance of medical electrical equipment and medical electrical systems".
Technical reports are also available for other standards, e.g., “IEC/TR 80002-1 Medical device software – Part 1: Guidance on the application of ISO 14971 to medical device software.”
This is a brief overview of the standards structure. If you have any further questions about electrical safety, we would be happy to assist you. For more information about 60601 and other medical technology topics, please read our other articles and visit our YouTube channel.
Best regards
Martin Bosch
