Are you looking for support with hardware development? Do you want to accelerate your project? Do you have a resource bottleneck? Do you have a technical problem that you're stuck on?
We develop hardware for medical electrical devices that must meet the requirements of 60601-1 or 61010-1. In this article, we want to provide insight into how we work and how we can support you:
Tools in hardware development
We work on a wide variety of projects. Typically, such development follows a classic process and can be broken down into the following steps:
| Dipl.-Ing. Martin Bosch, shareholder, hardware developer E-mail: bosch@medtech-ingenieur.de Phone: +49 9131 691 241 |
|
Do you need support with the development of your medical device? We're happy to help! MEDtech Ingenieur GmbH offers hardware development, software development, systems engineering, mechanical development, and consulting services from a single source. Contact us. |
|
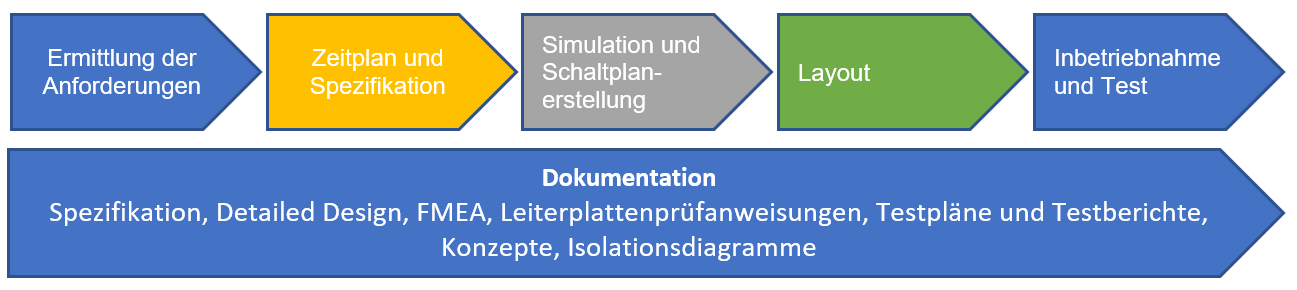
We primarily use Altium Designer for schematic design. As a schematic and layout program, Altium Designer offers us several advantages:
- Clear and simple library management.
- Easily integrated with our version control system Subversion.
- Export of 3D data in various formats possible.
- All the features we need, such as design rule check, routing of differential signals, length adjustment, etc., are supported.
The last two points are important for the EMC-compliant integration of printed circuit boards into an overall system. Medical technology, in particular, places high demands on immunity to interference, in addition to radiation. EMC-compliant design requires experience and proven methods, from concepts to layout and testing to approval.
We use LTspice for circuit simulation. Simulation models from other providers are not available.![]() They can be integrated in PSpice format, and data exchange with customers is easy because it's a free tool. It offers many features such as state machines, logic, and mathematical functions.
They can be integrated in PSpice format, and data exchange with customers is easy because it's a free tool. It offers many features such as state machines, logic, and mathematical functions.
Hardware-software codesign
Hardware development rarely exists in isolation. Solutions are typically developed within a system involving a system architect, software development, quality management, and engineering. We can provide excellent support here through close collaboration and our methodology.
Planning and costs
We create a schedule and cost estimate for each project.
Planning without execution is usually useless – execution without planning is usually fatal.
Willy Meurer
Measuring instruments, testing and commissioning
Hardware development also includes commissioning and testing. Our laboratory is equipped with essential measuring instruments such as oscilloscopes, multimeters, spectrum analyzers, and signal sources.
We'd be happy to support you with your hardware development with our experience and our experts, and perhaps we already have a solution to your problem. Feel free to contact us; we treat customer projects confidentially. We'd be happy to present our technical expertise to you in a personal meeting.
Best regards
Martin Bosch
