Failure modes and effects analysis (FMEA) is a risk management method. ISO 14971 states:
"Failure Mode and Effect Analysis (FMEA) and Failure Mode Effect and Criticality Analysis (FMECA) are techniques that systematically determine the effect or significance of individual components; they are more suitable for a more sophisticated design."
When used consistently, FMEA is an important tool in the medical device manufacturer's toolbox. FMEA can be used in a variety of areas, such as process FMEA (PFMEA) to evaluate failure modes in processes or design FMEA (DFMEA) to identify and treat defects in products.
In the development of medical electrical devices, it is typically used at the module level to analyze failure modes and define corrective actions. The best time to perform an FMEA is after the schematic has been created and before the layout, as changes can still be made at this point.
FMEAs are, of course, used not only in medical technology, but also in aircraft construction and the automotive industry. Various organizations provide guidelines and regulations for their implementation; among them, DIN EN 60812 deals with FMEAs.
Terms
The following table shows important terms related to FMEAs.
| German | English | Explanation / Meaning |
|---|---|---|
| ID | ID | Clear identification of the error (e.g. line number) |
| Component / Function | Item / Function | The FMEA refers to components or functions. |
| Possible error / malfunction | Potential Failure Mode | What kind of error can occur? |
| Cause of error | Failure Cause | What is the cause of the error |
| Error sequence | Effect of Failure | What does the error lead to (danger to patients, users or third parties, failure, etc.) |
| Preventive measure | Preventive Action | What is being done to prevent the error from occurring? |
| Discovery measure | Detection Action | What is being done to identify the error? |
| Rating (B) | Severity (S) | How big is the impact of this error? |
| Probability of occurrence (A) | Occurence (O) | What is the probability of occurrence? |
| Probability of detection (E) | Detection (D) | What is the probability of detection? |
| RPN (W x A x E) | RPN (S x O x D) | Risk priority number |
| Recommended remedial action | Recommended Action | What is the recommended remedial action? |
| Measure implemented | Actions taken | What is the implemented measure? |
| Rating (B) | Severity (S) | What is the impact of this error after the action? |
| Probability of occurrence (A) | Occuracy (O) | What is the probability of this error occurring after the measure? |
| Probability of detection (E) | Detection (D) | What is the probability of detecting this error after the measure? |
| RPN (W x A x E) | RPN (S x O x D) | Risk priority number after the measure |
Implementation
The FMEA is prepared and conducted in a group setting. Participants in a design FMEA include the module's hardware and firmware developers, the system architect, and possibly quality management staff.
The FMEA should be prepared in advance. However, sufficient time must be planned for its implementation. Depending on the size of the assembly, an FMEA can take from three hours to several days.
| Dipl.-Ing. Martin Bosch, shareholder, hardware developer E-mail: bosch@medtech-ingenieur.de Phone: +49 9131 691 241 |
|
Do you need support with the development of your medical device? We're happy to help! MEDtech Ingenieur GmbH offers hardware development, software development, systems engineering, mechanical development, and consulting services from a single source. Contact us. |
|
Proceed
The sequence of the individual error considerations is shown in the following diagram:
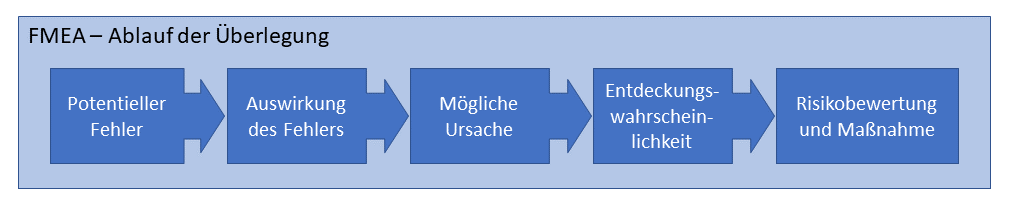
Results of an FMEA
When conducting an FMEA, various results and insights are obtained:
- Possible errors and failure modes of the module or system.
- Possible initial errors that endanger patient safety.
- Notes for necessary self-tests or runtime tests.
The findings from the FMEA can result in new requirements for the system.
Conclusion
Consider FMEA as a useful tool to improve the quality of your products.
If you would like to know more about how we conduct an FMEA, please feel free to write or call us.
Best regards
Martin Bosch
Further information on FMEAs can be found at:
[1] MIL-STD-1629A
[2] Wikipedia
[3] WW Vandenbrande, How to use FMEA to reduce the size of your quality toolbox, Quality Progress, 1998
