An important topic in our daily work is ensuring the electrical safety of the devices we develop. Other articles have already covered a lot about electrical safety and insulation diagrams (Part 1 and Part 2). In this article, I would like to discuss in more detail the various leakage currents, their significance for safety, and finally, the tests for leakage currents.
What is leakage current?
Leakage current refers to direct currents (DC) or alternating currents (AC) which:
- flow through the protective conductor.
- flow through the patient to earth.
- flow through the user to earth.
Direct currents can flow when several conductive surfaces with different potentials are touched simultaneously. These can be electrical devices, furniture, patient beds, or other conductive parts.
alternating currents can flow through parasitic capacitances. For the patient, a parasitic capacitance to ground is assumed in the range of 5–200 pF (see human body model: 150 pF in EN 61000-4-2 as a representative capacitance of the human body).
Devices can be designed so that leakage currents within the device are diverted to the protective conductor. In this case, they are referred to as Class 1 devices. However, there is a risk that higher leakage currents may occur in the event of a fault with an open protective conductor. In medical technology, this is referred to as a single fault condition. Leakage currents are also measured for various first faults, such as an open protective conductor, a broken conductor, mains voltage at the patient, and others.
Significance for human security
Leakage currents can have various adverse effects, including electrical stimulation, burns, or other injuries to the patient. Therefore, medical devices must meet strict safety standards to ensure electrical safety.
In medical technology, the patient is considered particularly in need of protection for several reasons:
- The patient can be connected to multiple medical devices at the same time.
- The patient may be unconscious.
- The patient may be sedated or anesthetized.
- The patient may have higher conductivity due to open wounds or during surgery.
As the above points show, protective reflexes can be eliminated.
The effect of currents on the body depends on
- Height of the current
- Duration of current flow
- Path of the current through the body (hand to foot or hand to hand, …)
At 230 V, an impedance of approximately 1000 Ohm applies to 50 % of the population at 50 / 60 Hz (hand to hand, with a large transmission area, see IEC 60479).
A current flow through the heart is particularly dangerous, since every current through the heart has a certain probability of causing ventricular fibrillation (see 60601-1, Figure A.14).
For general effects of electricity on the body, there are the well-known diagrams for the release threshold and flicker probability in electrical accidents:
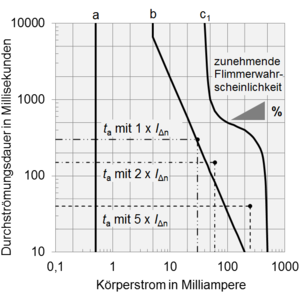
The following leakage currents are distinguished in 60601-1:
- Earth leakage current flowing from the power supply into the protective conductor.
- Touch current, formerly known as housing leakage current.
- Patient leakage current
- Depending on the classification of the applied part (e.g. applied part class BF 100 µA in normal case, 500 µA in the first fault)
- Mains voltage at the application part
- Application part to earth
- Mains voltage at other ports (signals, communications, supply, commonly referred to as SIP/SOPs)
- Mains voltage on conductive non-protective conductor connected housing parts
In addition, there are also patient auxiliary currents, for which limits are also defined in 60601-1. Patient auxiliary currents are currents that are required for a device's function and flow between patient terminals, for example, a current for impedance measurement.
Limit values for leakage currents in medical technology depend on various factors:
-
- the classification of the applied part.
- Is the device permanently installed or can the protective conductor be interrupted?
- Multiple sockets are treated separately and the current in the protective conductor of the multiple socket must be less than 5 mA.
| Limit values according to EN 60601-1 | ||||||
|---|---|---|---|---|---|---|
| Classification of the applied part | Type B | Type BF | Type CF | |||
| Device status | NC | SFC | NC | SFC | NC | SFC |
| Earth leakage current, device not permanently installed | 5,0 | 10,0 | 5,0 | 10,0 | 5,0 | 10,0 |
| Touch current | 0,1 | 0,5 | 0,1 | 0,5 | 0,1 | 0,5 |
| Patient leakage current | 0,1 | 0,5 | 0,1 | 0,5 | 0,01 | 0,05 |
| Notes | Table for AC All currents in mA NC: Normal Condition SFC: Single Fault Condition |
|||||
Measurement of leakage currents
Leakage currents can be measured using specialized measuring devices from manufacturers such as Fluke, Gossen Metrawatt, or Rigel. Currents can also be easily measured during development using a measuring device with an oscilloscope or true RMS multimeter.
The measuring device should have a bandwidth of up to 1 MHz. The input resistance should be 1 MOhm and the input capacitance should not exceed 150 pF.
The tests are conducted at 110 V/60 Hz, the highest rated mains voltage, and at the highest supply mains frequency. This means that if a device is rated for 115 V/60 Hz and 230 V/50 Hz, the tests should be conducted at 253 V/60 Hz.
Conclusion
Leakage currents must be considered during development, especially potential failure scenarios. Early consideration of failure scenarios in components, insulation, and ground connections ensures safe designs and reduced risks for users and patients. This article only outlines general requirements; please check the basic standard and the specific specifications for your device in each individual case.
If you have any questions or comments, please write to us or call us. We welcome feedback and are happy to discuss topics related to electrical safety in medical technology, especially your application.
Best regards
Martin Bosch
Sources, unless already mentioned in the text:
DIN EN 60601-1:2013-12
