1, 2 or 3 is not only the question in a well-known quiz show for children on public television, but also for companies that want to place a medical device on the market in the EU.
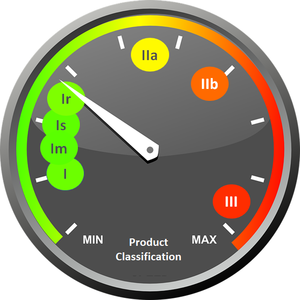
The MDR provides for classes I, IIa, IIb, and 3 in Article 51. In addition, three further classes come into play in Article 52 (7 a, b, and c).
- Is: for sterile products
- In: for products with measuring function and
- Ir: for reusable surgical products
Medical devices are classified using a risk-based system that considers the vulnerability of the human body and the potential risks associated with the devices. This approach uses a range of criteria to determine classification. These include, for example, the duration of contact with the body, the degree of invasiveness, the local or systemic effect, the potential toxicity, the part of the body affected by the use of the device, and whether or not the device is dependent on an energy source. These criteria can then be applied to a wide variety of medical devices and technologies.
These classification rules are set out in Annex VIII to the Medical Device Regulation (EU) 2017/745 (MDR).
To put it simply, the effort required for product approval increases with the higher the product risk.
In addition to Annex VIII of the MDR, there is now also a guide from the Medical Device Coordination Group. The MDCG 2021-24 Guidance on Classification of Medical Devices, published in October 2021, provides interested readers with detailed, 57-page guidance with clear decision-making rules and examples. I therefore highly recommend this document to anyone involved in product classification.
You can download the document at the following link.
https://ec.europa.eu/health/sites/default/files/md_sector/docs/mdcg_2021-24_en.pdf
Do you need support or advice in developing your medical device? Then please feel free to contact us. Our experienced MEDtech engineers will be happy to help you develop your medical device or clarify any questions you may have.
Best regards
Goran Madzar
