It's always interesting to think outside the box – how is hardware developed in other industries?
One of the most successful fields of engineering is aircraft engineering. According to the website planecrashinfo.com The number of accidents has decreased from the 1950s to the present, although the number of flights has increased significantly from 1970 to the present (source: gapminder.org). The aircraft engineers seem to be doing something right.
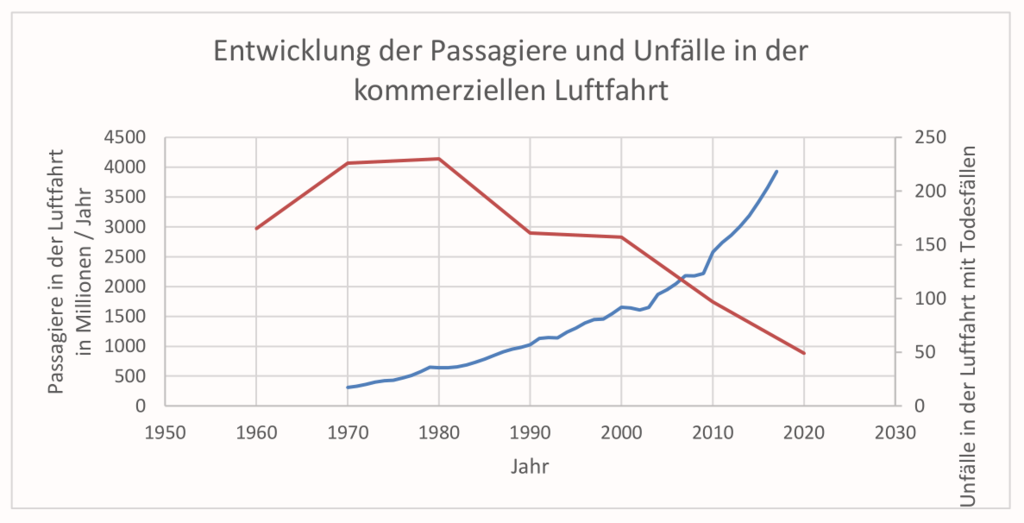
The reduction in aviation accidents and the increase in safety certainly have several causes, including strict regulation and high requirements for licensing, advances in training, the use of new technologies and the use of checklists (here our blog about checklists).
One aspect that contributes to the quality of electronics is the development processes. These processes are strictly regulated in aviation by RTCA DO-254 / EUROCAE ED-80, Design Assurance Guidance for Airborne Electronic Hardware. This standard is a process standard (like ISO 14971 or ISO 13485). This means that it does not explain how and what is tested; instead, it formulates requirements for the development process and its verification.
What is DO-254 about and what are the requirements for hardware development?
According to DO-254, hardware is divided into simple and complex systems. Simple systems can be fully tested and are not subject to the process requirements of DO-254. Complex systems cannot be fully tested, meaning their functionality and safety cannot be fully verified. This primarily includes systems implemented with programmable logic in CPLDs and FPGAs (hence the HDL-heavy description in the following chapters). To ensure that the system functions and is safe, a development process is necessary.
In DO-254, the HW development process is divided into 5 steps:
Recording of requirements
System requirements are captured and documented. The system architecture (and system-level requirements), including elements such as test structures and interfaces, are described and documented. Block diagrams, state diagrams, and flow charts are created that conform to the requirements.
Conceptual design
In this phase, the hardware design can begin with HDL development. The results of these activities are verified in reviews and, where possible, through automated testing (in HDL). Simulation is part of the verification and validation process, but also a component of the conceptual and detailed design process.
Detailed design
In this phase, the design is synthesized and the place-and-route process is completed.
In VHDL programming, bit streams are generated in this phase.
implementation
During this phase, FPGAs are programmed and prototypes are developed to enable testing and debugging.
Transition of production
In the final phase, the FPGA or board is prepared for release to production. After the board and FPGA are fully tested, production and production tests are validated. Acceptance tests and production quality tests must be defined.
In parallel, other processes such as verification and validation, certification, process monitoring and configuration management run.
What can we learn from this in medical technology?
In medical technology, unlike software, there are actually no requirements for the development process for hardware.
The procedure for software development for medical products is defined in IEC 62304.
HW development is excluded, as electrical safety for “more dangerous” devices with a higher safety classification is tested by a notified body in order to be approved for the market.
However, at MEDtech Ingenieur GmbH, we and other medical technology companies have also specified development processes for hardware development. In this development process, development projects follow a process very similar to DO-254, which is just as clearly structured. The project begins with requirements gathering, followed by a concept phase, the development phase, and finally, after validation and verification, the transition to production. Between the individual phases, there are gates at which certain documents must be submitted and reviewed. Following the V-model, we start at the system level and break this down into software, hardware, mechanics, and possibly other components.
We have had positive experiences with this process and can only recommend it.
Best regards
Martin Bosch
References: https://www.xilinx.com/support/documentation/white_papers/wp401_DO254_FPGA_Designer.pdf
