Contents
- 1. Introduction
- 2. Excitation signals
- 3. Electrodes
- 4. Limitations and sources of artifacts and errors in EDA measurements
- 5. Conclusion
- References

Factors influencing an EDA measurement
1. Introduction
In the previous blog post “Electrodermal Activity: Uncovering the Electrical Signals of Emotion and Physiology” I explained the basics of EDA. Among other things, I discussed the background and applications of EDA measurement, including signal properties and measurement methods. If you would like to read it again or haven't seen the first part yet, you can find it here. Blog post find:
This second part of the EDA blog series offers a thorough analysis of the numerous factors that influence an EDA measurement. We have already established that EDA is a physiological measure that reflects the change in electrical conductivity on the skin's surface in response to an external emotional stimulus. [1]The EDA can be used to measure sympathetic nervous system activity and emotional arousal. It is a useful tool for studying emotional reactions and stress in a variety of academic and therapeutic areas.
An EDA measurement system consists of two major functional units: a circuit for generating stimulation signals and an acquisition and processing unit for the output signals. The system can be further divided into sub-blocks such as electrodes, sensors, an oscillator, an ADC, and a signal processing unit. There are several variables that can influence an EDA measurement; some important ones are discussed in this article.
In addition to listing these factors, I also provide options regarding their variation and their specific effects. I conclude each section of this post with recommendations for varying these factors. The final section of this blog post lists limitations and sources of artifacts related to EDA measurements.
2. Excitation signals
Exosomatic skin conductance recordings are an important method for measuring EDA. A controlled electrical signal is the key prerequisite for this methodology. [2]An undetectable electrical signal must be applied to the subject's epidermis in a controlled manner to record the response signal. The nature and quality of these excitation signals (stimulation signals) can greatly influence the quality of the measurement. In this section, I will examine various effects caused by properties of the excitation signal.
2.1. Type of excitation
Both controlled voltage (CV) and controlled current (CC) can be used as external excitation sources for EDA measurements, so I will list some of their properties and the important advantages and disadvantages here:
- Voltage application
Implementing a constant voltage source is much simpler, and no further output conversion is required, as the skin conductance of the output can be determined directly. Any modern microcontroller can deliver a voltage stimulus with the desired frequency and amplitude. As mentioned earlier, the sweat glands in and beneath the epidermis can be represented as a parallel network of resistors and capacitors, whose resistance (reactance) can vary depending on the hydration of each channel. When a voltage is applied to such a parallel network, the same voltage drops across the network, while the current flowing through these channels changes according to Ohm's law. On the other hand, a very low voltage can cause a large amount of current in the low-resistance channels, which can lead to changes in skin conductance that are not associated with the sympathetic nervous system. This can lead to erroneous results during analysis. Using voltage as an excitation signal can lead to polarization of the electrodes, which adds artifacts to the measured signal. An additional transimpedance amplifier (TIA) is required to convert the current output signals into voltage for analysis. - Power feed-in
By injecting current, the current flow through the electrodes can be controlled, preventing polarization of the electrodes. Varying the skin impedance as a function of the controlled current density increases the linearity of the measurement. All modern EDA sensors provide both EDL and EDR data, making the conversion from impedance to admittance quite simple.
The only major problem with using the CC signal as an external stimulus begins with the fact that some of the epidermal ducts have high resistance (dryer) compared to the well-filled ducts. In such a case, the low-resistance ducts must carry an unphysiologically high amount of current. This high current can cause biophysical effects on the skin (electroosmosis). This leads to a high nonlinearity in the measurement process. In addition to the nonlinearity, such a high current can damage the sweat ducts and harm the test subject.
With the controlled current source, no additional TIA is required, but an AC-coupled amplifier is needed to convert such weak voltage signals to higher signal levels. Furthermore, using a CC excitation source allows both endogenous and exogenous EDA measurements to be performed in a single system. [3]However, this arrangement requires a slow, time-varying alternating current to avoid interference. - Remarks
In most cases, the EDA sensor is used in conjunction with other vital sign sensors. Therefore, the choice of excitation mode also depends on the design of the peripheral circuits. Numerous analyses of measurements with both excitation types can be found in the literature. A detailed study is presented in [4] presented a nearly simultaneous measurement of EDA using CV and CC sources. For the DC measurement, the correlation coefficient is reported to be low at 0.67. However, when a low-frequency (100 Hz) signal is used as electrical stimulation, the results are highly congruent, and the correlation between AC-CC and AC-CV approaches 1 (the correlation coefficient is 0.96 to 0.99).
The study also concludes that the type of excitation signal is almost irrelevant for AC measurement. - Recommendation
Any mode can be used for low-frequency AC excitation, but a constant voltage is recommended for the safety of the test subject. While voltage can lead to nonlinearity in the measurement because the current density under the electrode cannot be controlled, this is no longer a problem with modern sensors and data processing techniques.
2.2. Method of excitation
In addition to the type of excitation, the method of excitation signals is also important. Should a unipolar constant electrical signal or an alternating current signal with a constant peak-to-peak value be chosen? I would like to address and answer these questions in this section.
- DC method
The DC method has always been the most widely used method for EDA recordings because both the system and the circuitry required are very simple. The constant unipolar voltage is the excitation method that has always been recommended by the Society for Physiological Research [2]The recommended voltage for such a measurement is 0.5 V, which represents a reasonable compromise between signal-to-noise ratio and measurement linearity. This method makes the conversion from resistance to conductance (or vice versa) quite simple, since the inverse of one yields the other. Although the direct current method is the most standardized and widely used technique, the problems associated with this method are immense. As mentioned earlier, the epidermis has complex electrical properties (both R and C). With direct current excitation, we can only record the resistive properties of the skin.The capacitive effect of the skin in the recording of EDR has only a very small variation and significance [4] Another problem with direct current excitation is the crossover of endogenous and exogenous signals, since the skin potential and the result of external excitation are inherently the same. At higher direct current levels (0.5 V), this effect can be largely minimized. The biggest problem with direct current signal excitation is the polarization of the electrode and skin.
To avoid polarization of the electrodes, the use of non-polarizing Ag/AgCl electrodes is recommended. However, it should be noted that these electrodes will also become polarized after a certain time. [5]For portable applications, Ag/AgCl electrodes are not always suitable and other electrodes may also have a higher polarization potential, which distorts an SC signal [6]The application of a direct current over a prolonged period of time leads to polarization on the skin surface, which can cause itching and irritation in the subjects.
- AC method
The AC method solves almost all problems associated with DC excitation. The effect of skin capacitance is now part of the measurement, and skin admittance and impedance are complex conjugates and not reciprocal. If a slowly changing signal with a low peak-to-peak value is applied, the resulting current is out of phase. This phase can be measured using lock-in amplifiers. [7]Nonlinearity and polarization effects are not important with alternating current excitation. Endosomatic and exosomatic measurements can be distinguished because they are different in nature. Measuring skin susceptibility is also possible in this case. With the AC method, a weaker signal (100 mV instead of 0.5 V) can be applied, which reduces nonlinearity effects due to changes in epidermal properties. - Remarks
The superiority of AC excitation over DC excitation has long been recognized. However, the complexity of the required circuits and the lack of research and empirical data meant that the DC method remained relevant. However, with the existing range of sensors, complexity is no longer an issue. Studies with experiments and data are also available to standardize the AC method. It is assumed that there is a negligible difference between the measured DC conductance (GDC) and the AC conductance (GAC). [7]. - Recommendation
For this purpose, I recommend using a very low-amplitude AC source. The selected amplitudes for the excitation signals can range from 30 mV to 100 mV. Such low-amplitude excitation enables EDA measurements without causing electroosmosis of the stratum corneum. The overall frequency of this signal should be kept very low; I will provide a detailed overview of frequency in the next section.
2.3. Frequency range for excitation signal
We've already seen that using AC pulses instead of DC excitation facilitates EDA measurements. The next question is what the optimal frequency or the best possible frequency range is.
All biomaterials are considered predominantly capacitive. At low frequencies, the susceptance caused by the capacitive nature of the skin is dominated by free ions. If the excitation frequency is too high, dielectric losses begin to dominate the SC response. The source of these losses are ions with limited mobility. If the excitation frequency exceeds the relaxation time of these ions, they begin to oscillate, leading to dielectric losses. If the frequency is too high, these losses dominate, and the effect of the SC is distorted or may be lost altogether.
The penetration depth of an excitation signal is a direct function of the frequency [5]At very high frequencies (in the range of a few kHz), measurements are dominated by deeper viable tissue rather than the stratum corneum. To maintain the dominance of the stratum corneum during the measurement, a low-frequency signal is therefore preferred.
The only disadvantage of a low-frequency excitation signal is that it limits the sampling frequency. However, considering the advantages of an AC excitation source, this seems like a reasonable compromise.
There is very little literature specifically addressing the effects of frequency on EDA measurement, however, there are some works such as [5] and [7] report on a fixed frequency range that can be used.
A low frequency signal with a range of 5-100 Hz is reported in [5]. While [7] Data from EDA measurements for three discrete frequencies, 8 Hz, 20 Hz, and 88 Hz, have been reported. However, none of these studies present a mathematical model for the relationship between frequency and the degree of attenuation for EDA signals.
Engineers from Analog Devices Inc. presented a paper at the COSMOL conference proposing a model for EDA recording with respect to frequency [8]In this work, a parameter "k" is defined as the degree of pick-up, i.e., the normalized change in current caused by skin moisture. This parameter determines the sensitivity to changes caused by sweating. This study is conducted on a forearm model and refers to dry electrodes, which are required for wearable devices.
The relationship can be stated as follows:
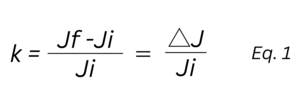
After performing a frequency sweep over a wide range (100 Hz–200 kHz), this paper reports that the best EDA measurement is recorded at 100 Hz. A significant increase in "k" is reported as the frequency decreases.
- Recommendation
A frequency of 100 Hz is recommended for low-amplitude AC signals. Another suggestion is 88 Hz, which avoids the second harmonics of the power grid (50 Hz).
3. Electrodes
Signal acquisition is the first and most important step in any measurement method; with biosignals, signal acquisition is significantly more difficult due to various factors. EDA recording is no exception. EDA recordings are typically performed using a bipolar two-electrode system, meaning two electrodes are applied to active skin sites.
The very first stage of signal acquisition is the electrodes, so factors such as the type and size of the electrodes as well as the distance between the electrodes play a crucial role.
The effects of all these factors will be briefly examined based on available studies. Unfortunately, there is a lack of high-quality studies and mathematical models based on empirical data in this area. Therefore, I will make electrode suggestions for you based on the best available results.
3.1. Size of the electrodes
The size of the electrodes is a direct measure of the contact between the skin and the electrode interface. The rule of thumb is that the larger the electrodes, the better the measurement. But unfortunately, this isn't entirely true without certain caveats.
According to the study by Mahon [6] It suggests a monotonically linear relationship between electrode size and SC signal strength. The relationship is somewhat more clearly linear for the slowly varying tonic component (SCL) than for the SCR (phasic component). Larger electrodes help minimize the possibility of electrolyte paste (or gel) seeping beneath the electrodes.
When using gel or wet electrodes, the problem of seepage is more likely to occur at a smaller electrode-skin interface. Even if seepage is present, the effect of seepage increases with a decrease in the area of the electrode-skin interface.
The first disadvantage, which occurs with increasing electrode size, is that the habituation effect increases at almost the same rate. Increased habituation leads to inaccurate EDA measurements over time. [6]Another problem is the adjustment period for the electrodes.
Each electrode needs time to establish better contact with the skin and the electrolyte content of the sweat. Before this time, EDA measurements are unreliable. The larger the electrode size, the longer this settling time. All these factors should be considered when deciding on the electrode size.
- Recommendation
For a better interface between electrode and skin, an area of 1 cm² is suggested [5]. Analysis of our data shows that this minimizes measurement error and results in minimal settling time and habituation.
3.2. Distance between the electrodes
This is another controversial factor in EDA measurement. Placing the electrode close enough eliminates the effect of skin potential, but the risk of short circuiting during heavy perspiration may increase.
Like the frequency of the excitation signal, the distance between the electrodes also affects the degree of SC acquisition. Fortunately, the same mathematical model that defines the relationship between excitation frequency and the degree of EDA acquisition can also be used to describe the effect of the electrode distance.
In the above-mentioned document, empirical data for electrode spacings ranging from 0.5 to 5 cm were analyzed, and the results indicate that the best results are achieved at a spacing of 0.5 cm (at a frequency of 100 Hz). It is noteworthy that the influence of the electrode spacing is not as dominant as that of the excitation frequency. The increase in the parameter "k" mentioned in equation (1) changes by only 5% when the electrode spacing is reduced.
- Recommendation
Although the distance between the electrodes depends on the specifications of the overall design, a distance of a few millimeters is recommended.
3.3. Type of electrode
Like any other measurement technique, the EDA recording method has evolved over the years, as has the selection of electrodes. To better understand the effects of the different electrodes, the electrodes are divided into two categories. A rough classification of the electrode types is shown in the following figure. [5].
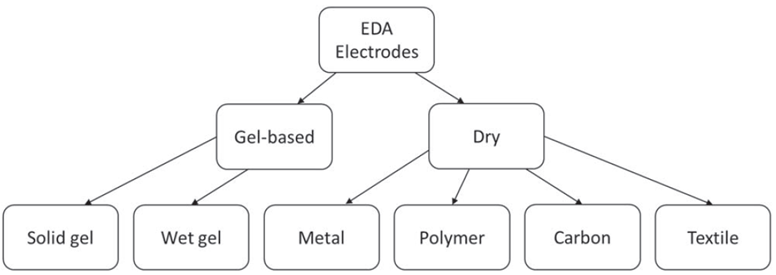
Figure 1: Classification of electrodes for EDA measurements [5].
- Wet gel electrodes
A moist gel or cream is applied to the electrode-skin interface to improve contact and charge transfer. It is a good measure to increase adhesion and reduce the interface impedance. The electrolytic properties of these materials are very similar to those of sweat. The problem with such electrodes is that they are not suitable for wearable applications like ours. Furthermore, moist gels or creams are more susceptible to pressure artifacts. Long-term use of these electrodes can lead to skin irritation in the subject. - Solid gel electrodes
As the name suggests, these electrodes use a solid gel with similar electrolytic properties instead of a paste or wet gel. These electrodes are mechanically more stable and provide more stable measurements, but have a longer settling time than wet electrodes. Electrode gel (both solid and wet) can alter the electrical properties of the skin when in contact for long periods. This property makes such electrodes less suitable for wearable applications, and durability is another concern. - Dry electrodes
The use of dry electrodes presents several new challenges compared to gel electrodes. Lack of adhesion leads to the formation of air pockets between the electrode and the skin surface, which can result in a higher impedance in series with the skin resistance. These air pockets are also the main source of pressure artifacts. Another challenge with a dry electrode is that charge transfer is more capacitive due to the lack of electrolytes to facilitate ionic charge transfer. This property makes SC measurement more a function of excitation frequency, which is not the case with gel electrodes. However, the absence of electrolytes increases the settling time compared to gel electrodes. Dry electrodes are ideal for wearable devices because their performance is consistent. Therefore, they are perfect for long-term EDA recording. Furthermore, without the electrolytic gel, there is no risk of irritation or itching.The table shows the results of a short study on different dry electrodes in comparison [5]:
| Metal electrodes | Polymer electrodes | Carbon electrodes | Textile electrodes |
|---|---|---|---|
|
|
|
|
4. Limitations and sources of artifacts and errors in EDA measurements
Recorded signals that are not part of a psychophysiological effect can be referred to as artifacts. In portable devices, the presence and effects of these artifacts are more pronounced than in a controlled laboratory environment. Some of the causes of these artifacts and errors are listed below:
4.1. Restrictions related to EDA
While EDA is a valuable measure of emotional arousal, it's important to note that it doesn't directly measure specific emotions. A high skin conductance can indicate both positive arousal and negative stress. The development of hardware for biocircuits has always been fraught with various challenges.
Furthermore, the inclusion of multiple biosensors in a single design and the unique properties of SCs complicate circuit design. Regarding signal (data) processing, interpreting EDA data inevitably requires careful consideration of the measurement context.
4.2. Sources of artifacts and errors
- Physiological sources
Speech activity, deep breathing, breath holding, and any other change in breathing pattern can induce EDA. In most cases, this causes changes in the rapidly changing EDR [5]. The EDR caused by these activities is at the discretion of the examiner and can be addressed during the signal processing phase of the EDA data. - Recording-based artifacts
The main source of such errors is the electrode-skin interface. As previously stated, these artifacts depend on the choice of electrodes. Electrode detachment, leading to the formation of air pockets, causes a high impedance in series with the SR. Motion and pressure artifacts are other sources of error. Applying pressure to human skin leads to a reduction in sweating and thus an increase in the SR. In addition, the electronic circuits of the measurement system can also introduce errors. These other artifact sources include noise generated by the signal generation and acquisition circuitry, as well as errors related to the analog-to-digital converter (ADC) unit. [2]. - Environmental sources
Temperature fluctuations at the recording site and the override of thermoregulatory sweating can cause errors that fall into this category. Sweating due to the dominant thermoregulatory response is caused by changes in ambient temperature and humidity. - Sources of noise
Noise at the electrode-skin interface, body noise, and ambient electrical noise are all sources of noise. As in any other case, noise at the electrode-skin interface is naturally the dominant source. Body noise such as ECG and EMG can also have an influence. A 50 Hz mains power supply can also contribute to EDA. [8].
5. Conclusion
In this blog post, I have largely explored all the variables that must be considered when designing an EDA measurement system, as they could impact design decisions. Other factors, such as electrode placement, medication side effects, ethnicity, gender, and age, have not been addressed here.
While all of these variables have only a small impact on the overall EDA score, they do have an impact. Most of the omitted variables are subject-specific and do not influence the design selection. While electrode placement is an important factor, with the advent of wearable medical devices, the choice of electrode placement has been limited to the area around the wrist.
This blog post focuses exclusively on the "Exosomatic" method of EDA measurement. It represents a step toward developing an EDA measurement system with minimal errors and artifacts. Efficient handling of artifacts and errors is the result of selecting improved signal processing techniques and hardware-level precautions.
The circuit topology is determined by the mode and type of excitation signals. These factors determine which combination of AC/DC voltage or current is used as the external electrical stimulus. The optimal excitation signal strength minimizes the effects of body noise such as EMG and ECG. Noise from external sources can be reduced by careful selection of the excitation frequency. The thoughtful and informed choice of electrodes is another factor contributing to accurate EDA measurements.
References
[1] Boucsein, Wolfram. Electrodermal activity. Springer Science & Business Media, 2012.
[2] Society for Psychophysiological Research Ad Hoc Committee on Electrodermal Measures, et al. “Publication recommendations for electrodermal measurements”. Psychophysiology 49.8 (2012): 1017-1034.
[3] Grimnes, Sverre, et al. “Electrodermal activity by DC potential and AC conductance measured simultaneously at the same skin site.” Skin Research and Technology 17.1 (2011): 26-34.
[4] Schaefer, Florian, and Wolfram Boucsein. “Comparison of electrodermal constant-voltage and constant-current recording techniques using the phase angle between alternating voltage and current.” Psychophysiology 37.1 (2000): 85–91.
[5] Tronstad, Christian, et al. “Current trends and possibilities in the methodology of electrodermal activity measurement.” Physiological Measurement 43.2 (2022): 02TR01 .
[6] Mahon, Mary L. The effect of electrode size on electrodermal measurement. Diss. University of British Columbia, 1986
[7] Pabst, Oliver. “Electrical properties of human skin: From linear recordings of exogenous electrodermal activity to non-linear memristor measurements.” (2018).
[8] Frequency and electrode spacing recommendations for EDA measurements (comsol.com)
[9] Greco, Alberto, et al. “Skin admittance measurement for emotion recognition: A study over frequency sweep.” Electronics 5.3 (2016): 46.
