Electrical fuses are critical components for the electrical safety of medical electrical equipment (ME equipment). They protect the device from incorrect operation, connection to excessively high or reversed supply voltages, and protect the operator and patient in the event of electrical short circuits within the device.
This article explains the different types of fuses and the requirements for fuses in medical electrical devices.
Types of fuses
Generally speaking, fuses are classified according to the following criteria:
- Working current
- Working voltage
- Melting integral (for fuses): I²t
- response time
- Tripping current
- Housing
- Approvals
The electrical properties and further information can be found in the respective data sheet and from fuse manufacturers such as Littelfuse, Vishay, Schurter, Weidmüller and many others.
| Dipl.-Ing. Martin Bosch, shareholder, hardware developer E-mail: bosch@medtech-ingenieur.de Phone: +49 9131 691 241 |
|
Do you need support with the development of your medical device? We're happy to help! MEDtech Ingenieur GmbH offers hardware development, software development, systems engineering, mechanical development, and consulting services from a single source. Contact us. |
|
Requirements for fuses in ME equipment
The following requirements apply to medical devices according to 60601-1:
- Each conductor in the supply of medical electrical equipment of protection class I or protection class II with functional earth connection must be provided with a fuse or an overcurrent release, except for the neutral conductor in permanently connected equipment and if two protection measures for all parts with opposite polarity are provided in the power supply (for details see 60601-1-8.11.5).
- No fuse may be provided in the protective conductor.
- The minimum requirement for the clearance and creepage distance before the fuse is a protective measure for operator protection between parts of opposite polarity in the power supply.
- Fuses must be dimensioned for the maximum current including current in case of fault and short circuit (see also note to 60601-1-8.11.5).
- A short-circuit test must be performed for the network between the device's own power supply (e.g., battery, accumulator) and the overcurrent protection, or two protective measures for operator protection (MOOP) must be provided. The requirement for the two MOOPs applies up to the fuse (see note to 15.4.3.5).
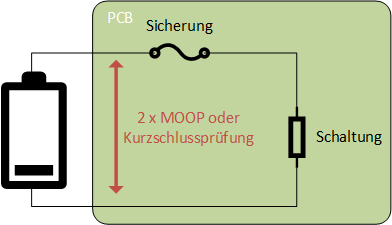
Requirements for clearance and creepage distances in front of fuses - There are special requirements for fuses in transformers.
- For replaceable fuses, the type and rating must be indicated next to the fuse holder, e.g., "T 315 L, 250 V." If a tool is required for replacement, information can also be provided in the accompanying documentation.
Conclusion
As a general rule of thumb, fuses should be located as close to the input as possible, electrically speaking. This "as far forward as possible" should also be taken into account in the layout to avoid violations of clearance and creepage distances.
Fuses are often critical components, so they should be included in the list of critical components and tested according to IEC standards. If possible, the certificates or verifications should be available or available from the manufacturer. UL or VDE certification with production monitoring is also advantageous.
In any case, you should carefully read the relevant sections of the standard again and not rely solely on this article. Feel free to contact me with any questions or suggestions.
Best regards
Martin Bosch
