Why EMC testing is vital in medical technology
Imagine a patient lying in critical condition in the hospital. Suddenly, a visitor's smartphone rings – and the monitoring device suddenly displays errors or delivers corrupted data. A frightening thought, isn't it? EMC testing during the approval process prevents precisely such scenarios.
But what tests are necessary to obtain approval for a medical electrical device? Why are pre-compliance tests useful? And what's the easiest way to test your device? That's what this blog post is about.
Which EMC standards apply to medical devices?
To place a medical device on the market in the EU, it must comply with the MDR (Medical Device Regulation). This regulation sets out the legal requirements for the manufacturer. By affixing the CE marking, the manufacturer declares that their product complies with all relevant European legal regulations. EMC testing is part of this comprehensive process.
The fundamental standard for the test is IEC 60601-1-2. This standard specifies the requirements for immunity and emissions for all types of medical devices. It defines the test levels and frequencies that a device must withstand, as well as the limits for emissions that must be observed.
IEC 60601-1-2 can be supplemented by special provisions, depending on the type of medical device, e.g. 60601-2-4 Chapter 202 for defibrillators.
Which EMC tests must medical devices pass?
The standards require a variety of tests, which can be divided into two main categories: immunity tests and emission tests.
category | standard | test | Description |
|---|---|---|---|
immunity | IEC 61000-4-2 | ESD | Electrostatic discharge |
IEC 61000-4-3 | (One) irradiated fields | Influence of high-frequency electromagnetic fields | |
IEC 61000-4-4 | Fast transients and bursts | Interference pulses are coupled onto network and data lines. | |
IEC 61000-4-5 | Impulse voltages | High-energy overvoltage impulses on supply lines | |
IEC 61000-4-6 | Conducted disturbances | High-frequency interference signals are coupled onto all cables. | |
IEC 61000-4-8 | Magnetic field | The device is exposed to a strong magnetic field at the mains frequency. |
Why are pre-compliance tests so important in EMC development?
Often, a product is initially developed without a comprehensive EMC analysis and then subjected to certification testing at the end. This frequently results in costly rework with makeshift solutions such as additional shielding, ferrites, and cable shields, or repeated, time-consuming test loops. This reactive approach is not only frustrating but also wastes valuable time and money.
Consideration of EMC requirements must begin as early as the initial system development phase. The major advantage: The costs for EMC measures are minimal in the design phase. (often only a few euros for additional components on the circuit board), but increase exponentially the later they need to be implemented in the development process.
The graphic by Henry W. Ott (1988) perfectly illustrates the core principle:
The later in the development process an EMC measure is implemented, the higher the associated costs. At the same time, the number of available solution options decreases drastically. Pre-compliance testing is key to identifying problems early, when solutions are still simple and inexpensive.
Our goal is to avoid this expensive and inefficient trial-and-error process. Instead, we are building on a systematic EMC development work.
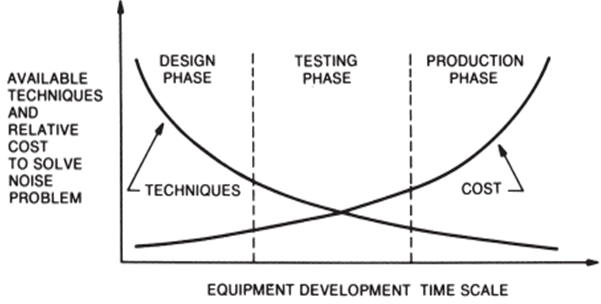
Figure 1: Figure 1-1 from Electromagnetic compatibility Engineering by Henry W. Ott
How we conduct EMC tests for medical devices
Laboratory measurements – pre-compliance tests
Before a device undergoes the expensive, accredited certification measurement, we carry out extensive testing. EMC testing during development through. This allows us to identify and resolve problems early, saving time and money. Our most important tools are with us.
Use of oscilloscope, spectrum analyzer and ESD simulator
The oscilloscope is our universal diagnostic tool. We use it not only for pure signal analysis, but above all to... The effects of disturbances and their countermeasures become visible to do this, for example, switching spikes from power supplies or the effectiveness of an RC snubber, as in this blog article: https://medtech-ingenieur.de/stoeraussendung-eines-batterie-ladereglers/.
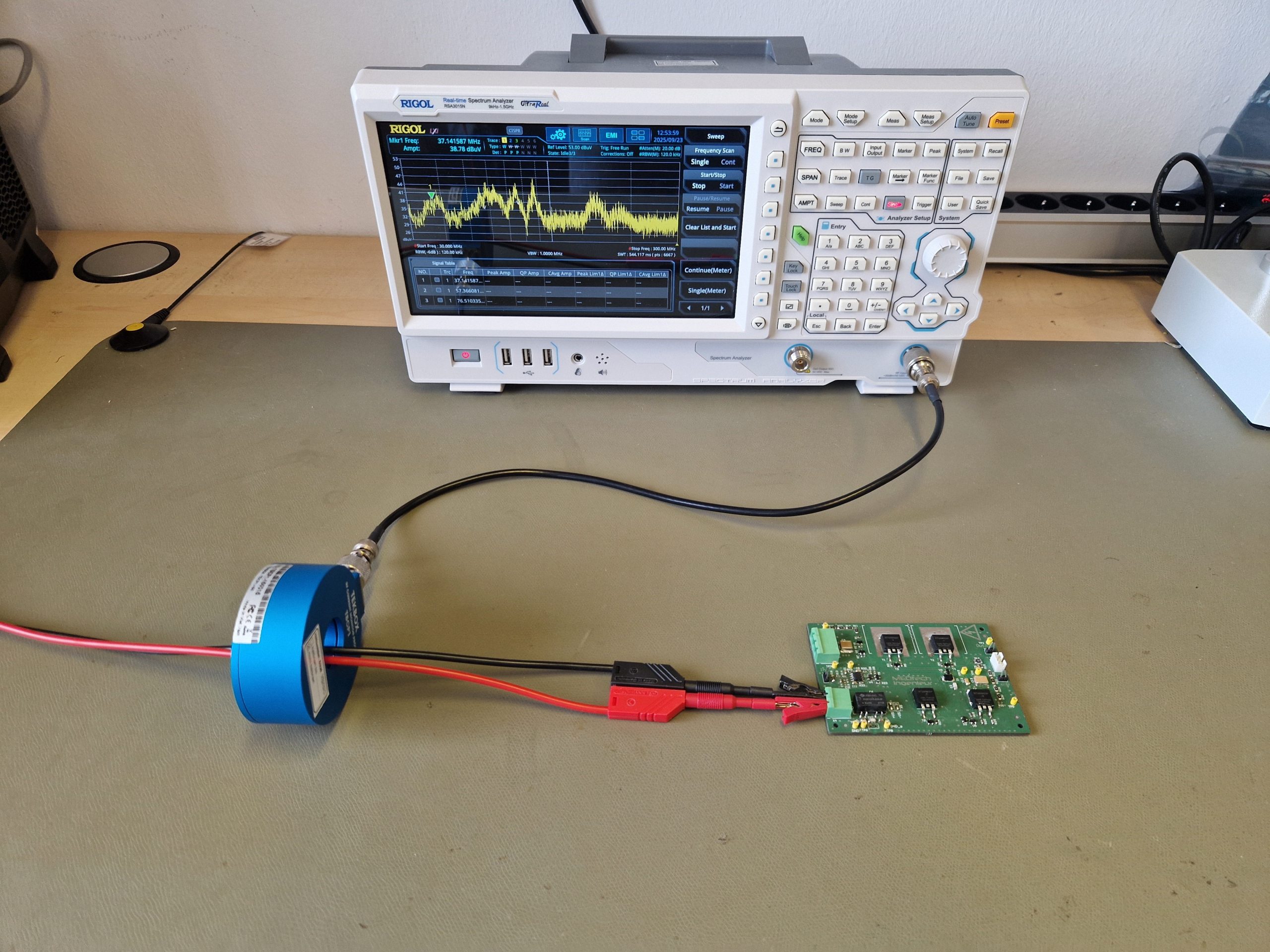
With the Spectrum analyzer We measure the frequency ranges in which a device emits radiation. This helps us develop targeted filtering measures, locate sources of interference, and make layout changes. You can find more information, for example, in this blog article: https://medtech-ingenieur.de/wie-behebt-man-emv-probleme/

Michael Wichert
Hardware developer at MEDtech Engineer
My practical tip: Measuring common-mode currents
Here is another example of a measurement that you should also perform yourself:
A current clamp allows you to quickly and easily measure the common-mode currents on your device's cables. These currents are often the cause of exceeding limits for radiated or conducted emissions.
By monitoring common-mode currents – ideally during development and with every layout change – many EMC problems can be identified early and specifically resolved.
ESD exposure can be critical for medical devices. With our ESD simulator We will check reproducible and controlled, how robust the circuit is against such discharges, where protective measures are needed, and verify their effectiveness.

EMC test chamber for realistic test conditions
The measurements with oscilloscope and spectrum analyzer take place in our laboratory (so-called). Pre-compliance testsFor more meaningful measurements of the radiated disturbances and immunity to interference radiation do we use a EMC test chamber. The measurement chamber for pre-compliance measurements is electromagnetically shielded and therefore virtually free from external interference. The measurement setup largely corresponds to that of a certification measurement and thus enables a meaningful and practical evaluation.
Early and development-accompanying measurements and our experience from numerous development projects and approval tests allow us to develop new circuits quickly and reliably.
Conclusion: EMC pre-compliance tests lead to faster, safe approval
For us as a development service provider, EMC is a consistent process. Quality and safety considerations, which is considered from the first circuit sketch to the final printed circuit board version.
Through EMC pre-compliance testing during the development of medical devices, we identify and resolve problems early on. This allows us to enter the final, accredited measurement with a device that is already optimized and pre-validated. saves costs, by implementing cost-effective solutions and reducing the number of test loops. This in turn significantly reduces the time-to-market.
Do you have a project where you need support or our EMC expertise?
Contact us!
